How Long Does It Take for a Baby to Withdraw From Subutex
A NIDA-supported clinical trial, the Maternal Opioid Treatment: Human Experimental Research (MOTHER) report, has found buprenorphine to exist a safe and effective alternative to methadone for treating opioid dependence during pregnancy. Women who received either medication experienced similar rates of pregnancy complications and gave birth to infants who were comparable on key indicators of neonatal health and development. Moreover, the infants born to women who received buprenorphine had milder symptoms of neonatal opioid withdrawal than those born to women who received methadone.
Methadone and buprenorphine maintenance therapy are both widely used to help individuals with opioid dependence attain and sustain abstinence. Methadone has been the standard of intendance for the past 40 years for opioid-dependent meaning women. However, interest is growing in the possible utilize of buprenorphine, a more recently approved medication, as another option for the handling of opioid addiction during pregnancy.
"Our findings suggest that buprenorphine treatment during pregnancy has some advantages for infants compared with methadone and is as safe," says Dr. Hendrée Jones, who led the multicenter report while at the Johns Hopkins University School of Medicine and is now at RTI International.
A Rigorous Trial Design
Methadone maintenance therapy (MMT) enhances an opioid-dependent woman'south chances for a trouble-gratis pregnancy and a healthy baby. Compared with connected opioid abuse, MMT lowers her risk of developing infectious diseases, including hepatitis and HIV; of experiencing pregnancy complications, including spontaneous abortion and miscarriages; and of having a child with challenges including low birth weight and neurobehavioral issues.
Along with these benefits, MMT may also produce a serious agin outcome. Like almost drugs, methadone enters fetal apportionment via the placenta. The fetus becomes dependent on the medication during gestation and typically experiences withdrawal when it separates from the placental circulation at birth. The symptoms of withdrawal, known as neonatal abstinence syndrome (NAS) include hypersensitivity and hyperirritability, tremors, vomiting, respiratory difficulties, poor sleep, and low-grade fevers. Newborns with NAS often crave hospitalization and treatment, during which they receive medication (oftentimes morphine) in tapering doses to relieve their symptoms while their bodies adapt to becoming opioid-complimentary.
The Female parent researchers hypothesized that buprenorphine maintenance could yield methadone's advantages for pregnant women with less neonatal distress. Buprenorphine, similar methadone, reduces opioid peckish and alleviates withdrawal symptoms without the rubber and health risks related to acquiring and abusing drugs. Therapeutic dosing with buprenorphine, every bit with methadone, avoids the extreme fluctuations in opioid blood concentrations that occur in opioid abuse and place physiological stress on both the mother and the fetus. However, dissimilar methadone, buprenorphine is a partial rather than full opioid and then might crusade less astringent fetal opioid dependence than methadone therapy.
The MOTHER study recruited women every bit they sought treatment for opioid dependence at six handling centers in the United states and one in Republic of austria. All the women were 6 to 30 weeks meaning. The research team initiated treatment with morphine for each adult female, stabilized her dose, and then followed with the daily administration of buprenorphine therapy or MMT for the remainder of her pregnancy. Throughout the trial, the squad increased each woman's medication dosage as needed to ease withdrawal symptoms.
The study incorporated design features to ensure that its findings would be valid. Among the most notable were measures taken to forbid biases that might arise if staff and participants knew which medication a adult female was getting.
To treat the participants without knowing which medication each woman was receiving, the study physicians wrote all prescriptions in pairs, one for each medication, in equivalent strengths. Report pharmacists matched the patient's name and ID number to her medication group and filled only the prescription for the medication she was taking.
Each day, participants dissolved 7 tablets under their tongues and and then swallowed a syrup. If a adult female was in the buprenorphine group, ane or more of her tablets contained that medication, depending on her prescribed dosage, while the rest of the tablets and the syrup were placebos. If a woman was in the methadone group, the syrup contained that medication in her prescribed strength and the tablets all were placebos. In this way, each adult female's complement of medications appeared identical to that of every other participant. The placebo tablets and syrup were crafted to wait, gustatory modality, and aroma similar the agile medications.
Equally Good For Mothers, Improve for Infants
Of 175 women who started a study medication, 131 connected until they gave birth. Those who received MMT and those given buprenorphine experienced similar pregnancy courses and outcomes. The two groups of women did non differ significantly in maternal weight gain, positive drug screens at nativity, percentage of abnormal fetal presentations or need for Cesarean section, need for analgesia during commitment, or serious medical complications at delivery.
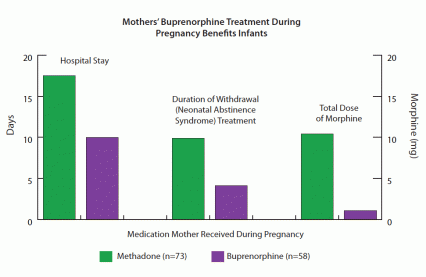
As the Female parent researchers had hypothesized, the infants whose mothers were treated with buprenorphine experienced milder NAS than those infants exposed to methadone (come across graph). Whereas most infants in both groups required morphine to command NAS, the buprenorphine group, on average, needed merely 11 percent every bit much, finished its taper in less than half the fourth dimension, and remained in the hospital roughly half every bit long as the infants exposed to methadone.
At Dr. Gabriele Fischer's Medical University of Vienna site in Austria, three women became pregnant for a second time during the fourth dimension Mother was enrolling participants. This development allowed researchers to compare the two medications' relative safety and efficacy in individual women as well as across groups. During her 2d pregnancy, each of the three women took the alternative medication to the one she took in her get-go pregnancy. In each instance, the child born following buprenorphine handling exhibited milder NAS symptoms than the one born following methadone treatment. This event suggests that differences in the effects of the two medications, rather than women's private differences in physiology, underlie the group findings.
"Buprenorphine may exist a good option for pregnant women, particularly those who are new to treatment or who become meaning while on this medication," says Dr. Jones. "If a patient is on methadone maintenance and stable, however, she should remain on methadone."
Next Questions
MOTHER researchers observed that although the women in their buprenorphine and methadone groups benefited as from treatment, the drop-out rate was college in the buprenorphine group (33 vs. 18 percent). This difference was not statistically significant. The researchers speculate that if information technology is meaningful, information technology may be owing to factors other than dissimilar responses to the two medications. They surmise that the experimental treatment protocols may have moved patients from morphine to buprenorphine too apace, causing discomfort, or that buprenorphine may accept been easier than methadone to discontinue when women decided to become abstemious.
The MOTHER study did not include women with some substance use disorders that are commonly comorbid with opioid abuse. "Time to come studies should compare neonatal abstinence syndrome, birth outcomes, and maternal outcomes of these ii medications for pregnant women who besides abuse alcohol and benzodiazepines," Dr. Jones says.
"The field also needs data on neonatal outcomes when pregnant women are treated with buprenorphine combined with naloxone, the electric current first-line form of buprenorphine therapy for opioid dependence," Dr. Jones notes. The MOTHER study administered buprenorphine without naloxone to avoid exposing the fetus to a second medication with potential agin furnishings.
"Research challenges remaining afterward this brilliant report are to determine the factors that resulted in the differential drop-out rates betwixt the ii medications," says Dr. Loretta P. Finnegan, who did pioneering work in the cess and treatment of NAS. "Additionally, researchers demand to acquit followup enquiry on these children to determine the longer term significance of the differences in newborn withdrawal symptoms." Dr. Finnegan, now president of Finnegan Consulting, was formerly the medical advisor to the managing director of the Office of Enquiry on Women's Health at the National Institutes of Health.
"Neonatal forbearance syndrome is a terrible experience for infants, and there is a great need to improve care for this condition," says Dr. Jamie Biswas of NIDA's Division of Pharmacotherapies and Medical Consequences of Drug Corruption. "Dr. Jones' study is a superb contribution to this surface area of clinical research, and the robust results should provide more treatment options for a syndrome that affects thousands of infants each year."
Sources:
Unger, A., et al. Randomized controlled trials in pregnancy: Scientific and upstanding aspects. Exposure to different opioid medications during pregnancy in an intra-private comparison. Addiction 106(7):1355–1362, 2011. Total Text
Jones, H.Due east., et al. Neonatal forbearance syndrome after methadone or buprenorphine exposure. New England Periodical of Medicine 363(24):2320–2331, 2010. Full Text
MOTHER Collaborators
Following is a listing of collaborators on the Maternal Opioid Treatment: Human Experimental Research (MOTHER) Written report and their university affiliations.
Dr. Hendrée Jones (written report leader), Johns Hopkins University Schoolhouse of Medicine; Dr. Amelia Arria, University of Maryland, College Park; Dr. Mara Coyle, Warrant Alpert Medical School of Brown University; Dr. Gabriele Fischer, Medical University of Vienna; Dr. Sarah Heil, University of Vermont; Dr. Karol Kaltenbach, Jefferson Medical College; Dr. Peter Martin, Vanderbilt University Schoolhouse of Medicine; Dr. Peter Selby, University of Toronto; and Dr. Susan Stine, Wayne State Academy School of Medicine.
How Long Does It Take for a Baby to Withdraw From Subutex
Source: https://archives.drugabuse.gov/news-events/nida-notes/2012/07/buprenorphine-during-pregnancy-reduces-neonate-distress
0 Response to "How Long Does It Take for a Baby to Withdraw From Subutex"
Post a Comment